
LABS
With 2,000 sq. feet of dedicated R&D space in our state-of-the-art facility in Rochester, MN, Nucleus RadioPharma has the capacity and ability to prioritize your radiotherapy or theranostic product development, regardless of the isotope.
Take your scientific advancement to market with a full-service CDMO
Nucleus RadioPharma is an end-to-end contract development and manufacturing organization (CDMO) partner dedicated to building the robust clinical and commercial supply chains for targeted radiotherapies that make radiopharmaceutical companies—and the innovators driving advancements—successful.
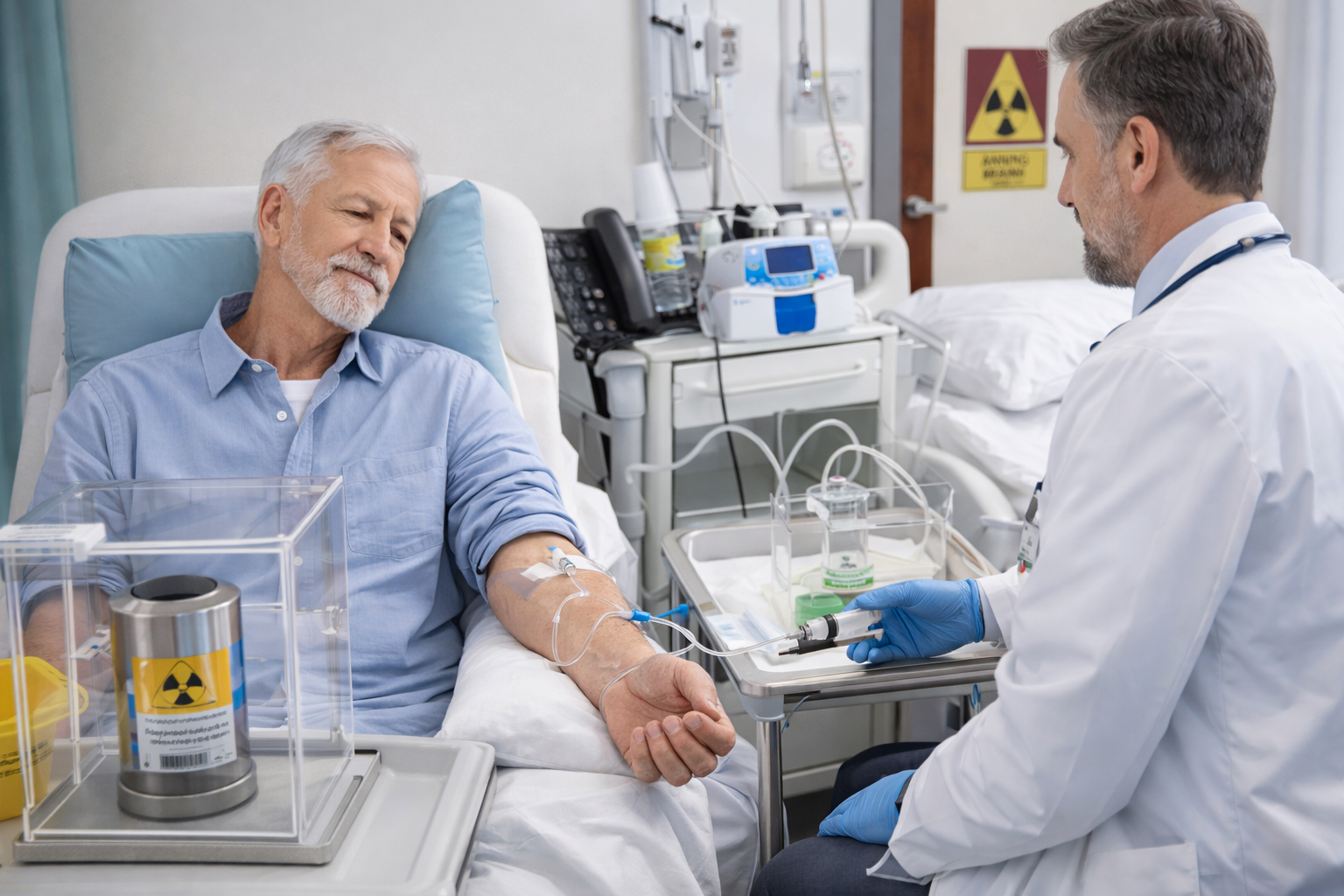
No matter where you are on your targeted radiotherapy journey, you’ll receive optimal services that rapidly advance your program—we’re your dedicated partner every step of the way, ensuring patients receive your targeted therapy
Dedicated R&D operations in MN
Dedicated Manufacturing Space in MN

With 2,000 sq. feet of dedicated R&D space in our state-of-the-art facility in Rochester, MN, Nucleus RadioPharma has the capacity and ability to prioritize your radiotherapy or theranostic product development, regardless of the isotope.
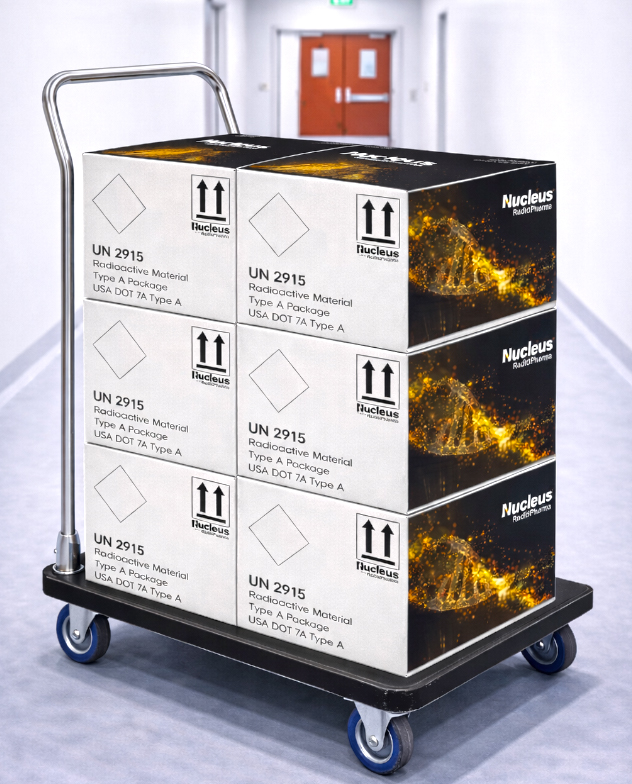
With an isotope-flexible approach and specialized radiopharmaceutical logistics expertise, Nucleus RadioPharma supports secure, reliable supply chain solutions across development and manufacturing programs.

With one fully operational space in Rochester, MN, and one expansion site under construction, Nucleus RadioPharma has the capacity and specialized infrastructure to meet any radiopharmaceutical manufacturing need—from clinical trials through commercial production.

With manufacturing facilities designed to support compliance with 21 CFR 211, Nucleus RadioPharma’s quality systems and regulatory approach are built to meet radiopharmaceutical safety, compliance, and operational requirements.
Every detail of our state-of-the-art, commercial-scale facilities is built with one purpose in mind: Advancing the development, manufacturing, and delivery of our clients’ radiotherapies quickly, efficiently, and safely.


Second-floor R&D labs are separated from first-floor GMP manufacturing, enabling parallel work, collaboration, and full GMP compliance.

Situated on a large life sciences campus, with over 47K sq ft of manufacturing space just north of Philadelphia, and conveniently located within an 8-hour driving radius of key markets

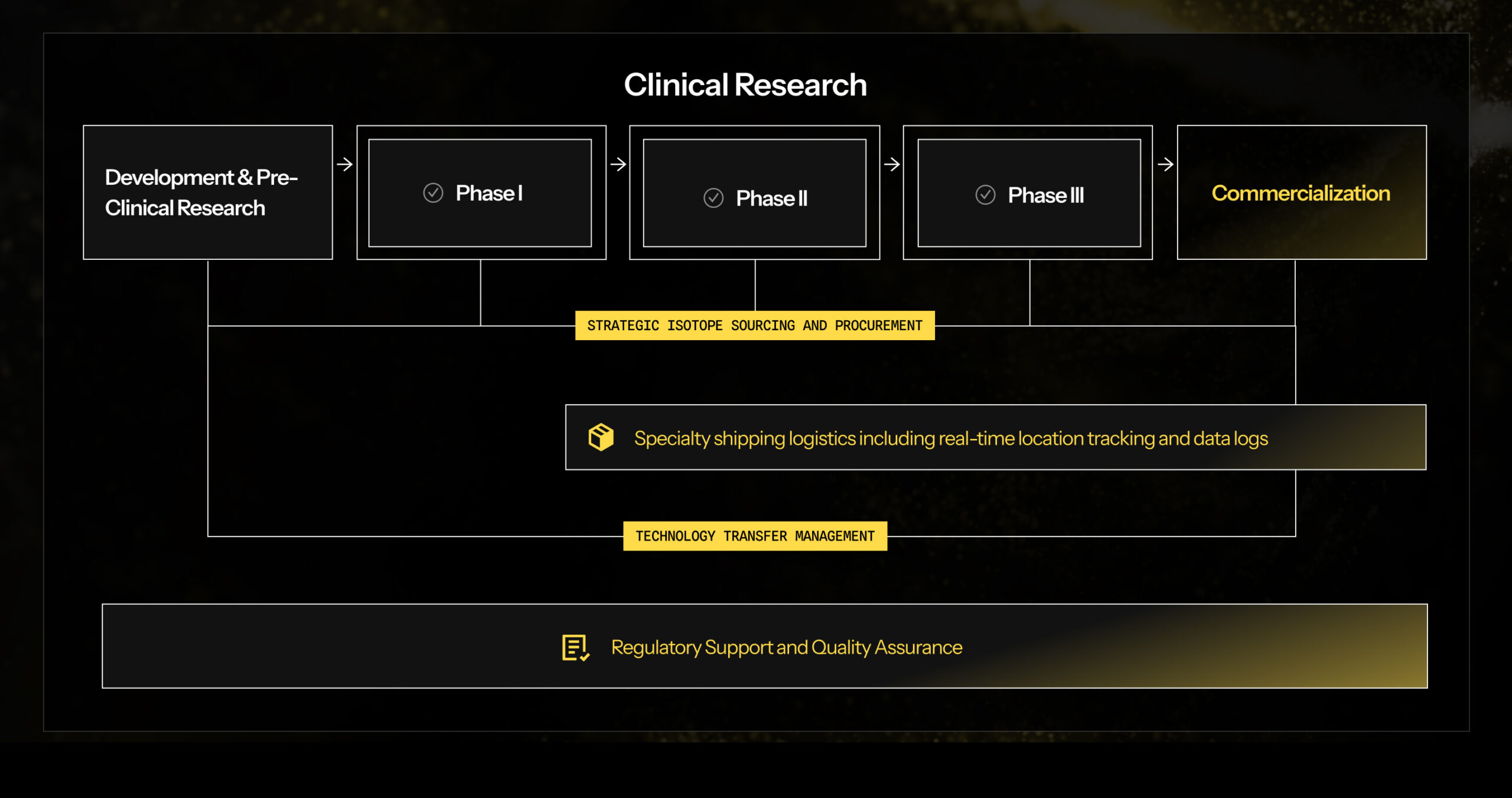


Curious about how our CDMO services can help your organization? Contact our Business Development Team to discuss our capabilities.
Contact UsExplore our open radiopharmaceutical jobs in Rochester, MN and Pennsylvania facilities.
